Our oceans and the life forms they support are under siege, threatened by a formidable collection of forces that cause both sudden mass mortalities and a slow degradation of biodiversity.
Big disease outbreaks are making our ocean biota sick, from corals with spots and halos to starfish melting away to salmon anemic with viruses.
Although top threats for our oceans are the warming and acidification that accompany climate change, there are other threats including over-fishing, pollution from land, and oil extraction. As a marine ecologist specializing in disease, I worry most about the threat posed by microbes, because in oceans beset by all these stresses, microscopic disease-causing organisms can gain the upper hand, cause death on a massive scale, and thereby bring about rapid, wide-scale ecological change.
The Danger of Microbes
Microbes are scary in part because they are changeable and not under our control. Pathogenic organisms in the microbe category—viruses, bacteria, fungi, protozoans, and other disease-causing agents that don’t fit neatly into these groups—are constantly evolving, their genetic codes often changing rapidly and staying one step ahead of their hosts’ defenses.
Think about one of the deadliest of human diseases, the Ebola virus, which causes fever, severe headache, vomiting, diarrhea, and hemorrhagic bleeding in its victims. Available evidence indicates that the virus has existed in bats in Africa for a long time, occasionally jumping to human beings but never breaking out beyond Africa.
Then in 2013 a horrific epidemic of Ebola virus started in Guinea, Liberia, and Sierra Leone, spreading faster and further in Africa than previous outbreaks. Declared a Health Emergency of Special Concern in August 2014, it ultimately killed over 11,000 people and reached Europe and North America.
Why was this outbreak so much bigger than earlier ones? Scientists aren’t sure, but one hypothesis, backed up by intensive study of the changing viral genome during the epidemic done by a team led by Daniel Park of Harvard’s Broad Institute, is that a key mutation allowed it to become more transmissible among humans. Because viruses have a very short life span and so many new virus particles are produced in the body of a single host, there is ample opportunity for such mutations to occur.

Microbes are dangerous too, because many can attack and infect more than one species or spontaneously develop that ability through a favorable genetic mutation. Pathogens that have a wide host range, called multi-host pathogens, tend to be deadly for at least some of the species they can infect. By deadly I mean they can kill every individual within a susceptible species, even driving them to extinction, while persisting in a more resistant host species.
Contagious individuals in a resistant species can keep exposing healthy individuals in a susceptible species until the susceptible species is wiped out. In these situations, we say that the pathogen has a reservoir in the resistant hosts—a nice comfortable hideout from which to spread. The starfish epidemic of 2013–14 was caused by a multi-host pathogen. It affected almost all the major species of the entire group we call starfish.
Those of us who study non-human diseases are worried that mass mortality caused by multi-host pathogens is becoming a common and recurrent event, threatening biodiversity in both terrestrial and marine environments. The frogs of the world’s rainforests are a high-profile example.
Many rainforest frog species have been not just devastated but completely eradicated, at locations around the planet, by a skin-attacking fungus, Batrachochytrium dendrobatidis, called chytrid for short.

This lethal fungus grows in the skin of the frogs, spreading its killing tendrils and exploding cells from the point of entry throughout the frog’s skin. It can kill some species within days and is wildly contagious.
In others, it lingers longer, so the infected frog continues to shed infectious spores from its skin into nearby streams. In 2015, using data from museum databases and field counts, John Alroy estimated the epidemic has caused the extinction of more than two hundred species of frogs.
The chytrid epidemic has happened so quickly and in such remote tropical locations that the exact number of extinctions is still unknown, but we do know that many species are forever gone from our planet.
Outbreak in the Caribbean
My career as a Disease Ecologist began in 1996, investigating a mass die-off of Caribbean sea fans on the tiny island of San Salvador, Bahamas. I first heard about these declines when one of my colleagues, Jim Porter, a professor at the University of Georgia, called me in Ithaca one day in 1994.
He’d heard at a meeting that sea fans were dying at many locations around the Caribbean. “If you need samples,” he told me, “this is the time to get them; your populations are disappearing quickly.”
In the following months, I communicated by phone and email with researchers who studied these animals and we all became unnerved by the growing reports of dying sea fans.

Given how widespread the die-off was throughout the Caribbean, I was worried about the impact it could have on sea fan populations. In the late 1990’s we launched a study in the Caribbean to document the ecology of this underwater epidemic and monitor the defensive function of the coral’s chemicals in nature.
I recount the story of this work in my new book, Ocean Outbreak, and how we eventually learned that the sea fans had super-powers to fight back.
Although sea fans and all the group we call corals are some of the most ancient body plans on our planet, they possess sophisticated immune systems with potent weapons against infectious disease.
This outbreak is one of the few catalogued successes of a coral population to suffer huge mortality, but eventually develop resistance and survive a large epidemic. The work with Caribbean seafans next led us to study health of corals in the coastal waters of Indonesia, the heart of coral biodiversity.


In the fall of 2013, we were still in the middle of studying the health of corals in Indonesia when starfish in nearby British Columbia began dying catastrophically. The paired photos above shows how quickly a large population of the sunflower star died over 3 weeks in October 2013.
I tell the more complete story in my book, but this outbreak continues into 2019 and that sunflower star, the most susceptible to a multi-host pathogen in 2013, is now severely imperiled along much of its range from San Diego to Alaska in both near shore and off shore deep water.
Connecting Human Health and Wildlife Health
There are many other examples of infectious diseases causing massive underwater outbreaks from oysters and clams to abalone to herring and salmon.
Climate change is making it worse, since a warming ocean can both stress hosts and make pathogens grow faster and therefore become more damaging. Despite the increase from a warming ocean, we understand little about ways that infectious disease outbreaks begin and are transmitted in ocean waters.
There are more knowledge gaps than answers. With so much uncertainty about both the causative agents and how they transmit in our oceans, it is tough to manage underwater outbreaks.
But we can capitalize on the things we do know: that conditions that are bad for human health are also bad for wildlife health. I describe in my book how managing for both human health and health of the ocean biota can help. There are natural disease-fighting services in intact marine ecosystems that can reduce risk for both humans and wildlife like corals.

The links between coral health and human health became clear to us when our team ran a coral health workshop on an island in the Spermonde Archipelago in Sulawesi, Indonesia. As we were leaving the island, we were all simultaneously sickened with either amoebic dysentery or typhoid.
One of our scientists had to be evacuated because she had both infections and was dangerously ill. We vowed to return to see what was in the water.
Two years later, Joleah Lamb (then a TNC Nature Net Postdoctoral Fellow collaborating with me and now Assistant Professor, University of California Irvine), returned and took water samples and sequenced all the bacteria.
She found a long list of sewage-derived human pathogens in the water adjacent to four of these islands. But even more interesting, she found that human and wildlife pathogens were reduced by half in seagrass beds near the islands.
Joleah found that not only were pathogenic bacteria halved inside the seagrass beds, but coral health was improved. I show her graph from our paper below, documenting big reductions in the number of corals with disease inside seagrass meadows.
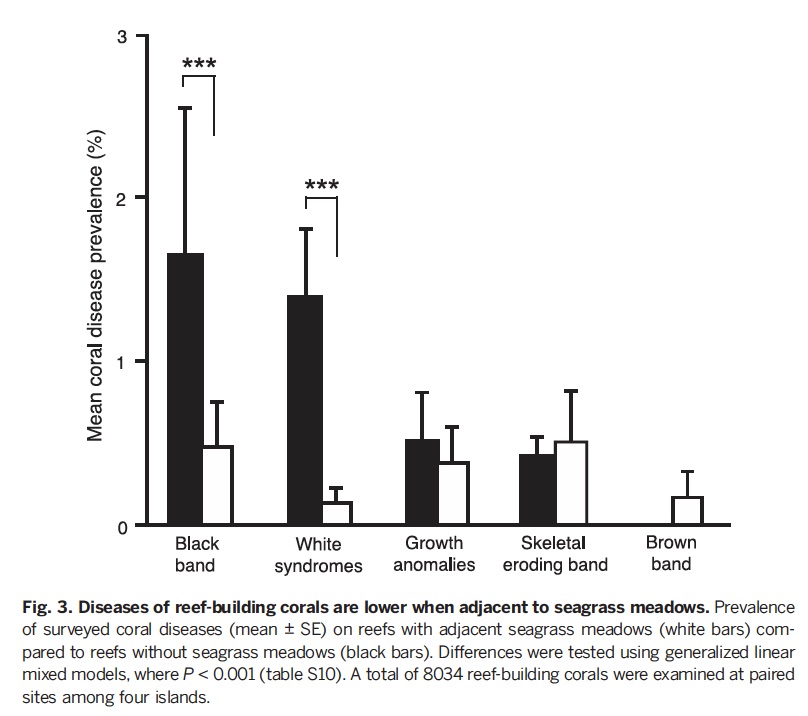
In those same seagrass beds, the levels of indicator bacteria are reduced by half when compared to adjacent non-seagrass sites near four separate islands. IN our paper, we also report the complete sequence data showing that many human and invertebrate disease bacteria are reduced in the seagrass beds
It is early days in this research program, but our goal is to look carefully at the ways that seagrass beds can reduce pathogenic bacteria. While we would never suggest that seagrass meadows be a replacement for state of the art septic systems, we do suggest that seagrass meadows (and other aquatic plants and intact ecosystems) are good backup hygiene for the ever-present spillover that does occur from many of our waste management systems on land.
Seagrass meadows may also have unrecognized ability to naturally reduce non-human pathogens of our ocean’s biota. Nature’s filtration and decontamination services can be particularly important for intermittent or unexpected introductions of new pathogens.
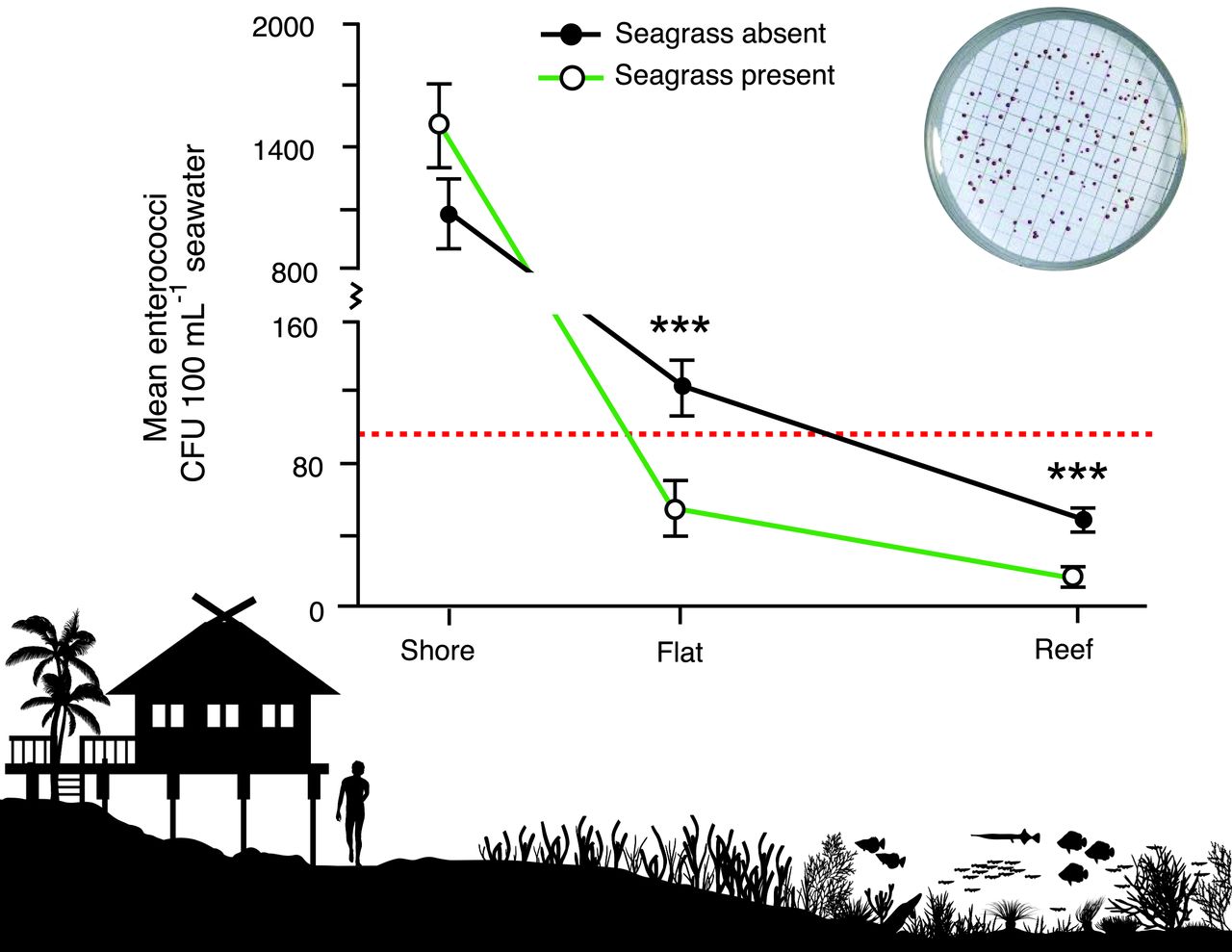
The observation that human and wildlife health are linked (as described in One Health) and can both be improved by better management of marine resources is a new approach that capitalizes on the value of natural filtration services of seagrass beds. This is only one of several new approaches to slowing a rising tide of new outbreaks I discuss in Ocean Outbreak.
Drew Harvell is Pa rofessor of Ecology and Evolutionary Biology at Cornell University and author of the newly released Ocean Outbreak: Confronting the Rising Tide of Marine Disease (University of California Press).




I never realized these problems were so prevalent and widespread. I knew we had ocean acidification and coral bleaching but nothing more than that. I never realized microbes were as much of a problem as they are but according to your content and pictures they are out there like anything else.
It is great to learn that sea fans have super-powers to fight back all this disease. I hope the Sea Star has the same powers.
I cannot quite take hold or comprehend the fact that human and wildlife health are linked but, that actually makes sense to me. Please keep me posted and keep in touch.
Alan
I want to thank everyone is involved in saving OUR EARTH. We have only one mother. We need to protect her. We HAVE to save it for children. THEY ARE OUR FUTURE. Do we kill or save ourselves? CHOOSE.